New Semi-Artificial Photosynthesis Study May Revolutionise Solar Power Storage
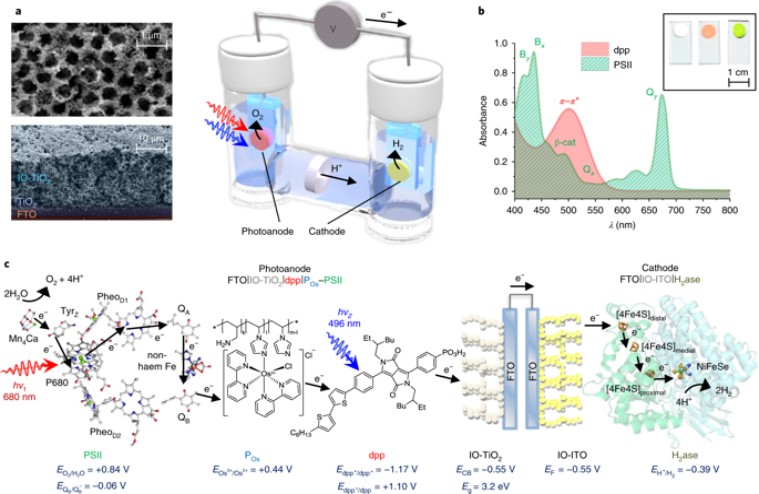
After the researchers successfully split the water molecules into hydrogen and oxygen by altering the photosynthesis process found in plants, the quest of harnessing solar power through different ways has got a new lease for further development.The latest study by academics of University of Cambridge led by St John’s College put semi-artificial photosynthesis to use to formulate a new strategy of producing and storing solar energy. The academics used a blend of modern technologies and biological components to convert water into hydrogen and oxygen from natural sunlight. One of the best outcomes from this study is that the methodology also resulted in more absorption of sunlight in comparison to the natural process of photosynthesis.
This unique process which can achieve unassisted solar-driven water-splitting is developed at the Reisner Laboratory in Cambridge and it is expected that the key findings of this research may now revolutionise the systems used for renewable energy.

Katarzyna Sokół, first author and PhD student at St John’s College, said: “Natural photosynthesis is not efficient because it has evolved merely to survive so it makes the bare minimum amount of energy needed – around 1-2 per cent of what it could potentially convert and store.”
Artificial photosynthesis has been around for decades but it has not yet been successfully used to create renewable energy. The issue of scaling process of renewable energy creation through artificial photosynthesis for industrial usage was marred by the fact that catalysts used in the process are often expensive and toxic. But the researchers at the Cambridge team instead used the enzymes to create the desired result thus overcoming the limitation of toxicity.
Researcher Katarzyna Sokol along with her team managed to reactivate a process in the algae that has been dormant for millennia. Researchers are now hopeful that innovative model systems for solar energy conversion can be developed through the findings.
She explained: “Hydrogenase is an enzyme present in algae that is capable of reducing protons into hydrogen. During evolution, this process has been deactivated because it wasn’t necessary for survival but we successfully managed to bypass the inactivity to achieve the reaction we wanted – splitting water into hydrogen and oxygen.”
Lead researcher Sokol asserted that the findings of this research can lead to the emergence of more robust solar technology.
The approach could be used to couple other reactions together to see what can be done, learn from these reactions and then build synthetic, more robust pieces of solar energy technology, she said.

This is first of its kind model to use hydrogenase and photosystem II for creating semi-artificial photosynthesis supported entirely by solar power.
Dr Erwin Reisner, Head of the Reisner Laboratory, a Fellow of St John’s College, University of Cambridge, and one of the paper’s authors described the research as a ‘milestone’.
He explained: “This work overcomes many difficult challenges associated with the integration of biological and organic components into inorganic materials for the assembly of semi-artificial devices and opens up a toolbox for developing future systems for solar energy conversion.”
Natural photosynthesis stores sunlight in chemical energy carriers, but it has not evolved for the efficient synthesis of fuels, such as H2. Semi-artificial photosynthesis combines the strengths of natural photosynthesis with synthetic chemistry and materials science to develop model systems that overcome nature’s limitations, such as low-yielding metabolic pathways and non-complementary light absorption by photosystems I and II.




